Hello to everyone. Today I have the personal pleasure to interview Dr. Grégoire Michoud. He is a friend and a brilliant scientist working on ecosystem microbial ecology. In the interview, Grégoire will talk to us about oil spills in the marine system, a specific anthropic hazard that can evolve into a natural hazard with terrible environmental consequences.

Grégoire Michoud is a Post-Doctoral Scientist at KAUST (Saudi Arabia), since 2015. He obtained his Ph.D. from the Université de Bretagne Occidentale, in Brest France. His primary research interest focuses on the characterization of the coping mechanisms of microbes in extreme environments (deep sea, brine pools, hydrocarbon pollution) by genomics and cultures approaches. His work has application in hazard chain from anthropic sources to natural targets.
Hello Grégoire, please tell us a bit about environmental issues related to oil spills.
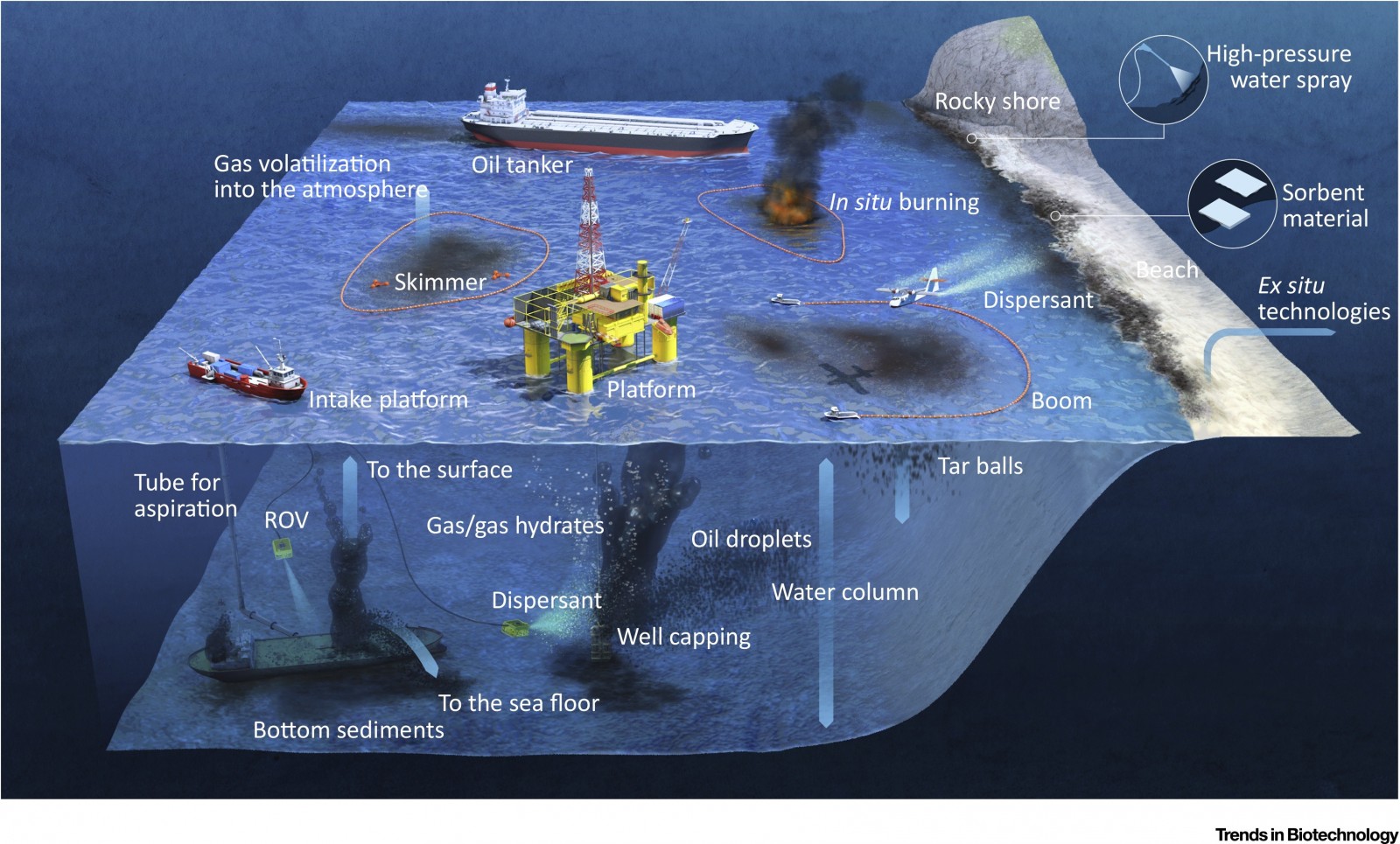
When it comes to oil spills, widespread publicized releases are rare events and represent just 10% of the overall release in the marine ecosystem. The remaining 90% is actually due to ship activities or other land-based industries that contribute to the pollution. One example of rare oil spill is the Deep Water Horizon case where, in a relative short time, a huge amount of oil was released into the Gulf of Mexico. However, in the vast majority of situations, small quantities of oil can be released from random events adding up to a considerable pollution.
What are the challenges when dealing with oil spills in the marine ecosystem?
I would like to point out that the environment itself has the ability to self-heal. In fact, over time the physical energy of the waves or the exposure to sunlight can break down the hydrocarbon compounds. Even some bacteria that are naturally present in the water column can digest the oil. Hydrocarbons are chains of carbon that can be structured in a very simple way or can be much more complex. The simple chains are easily and naturally degraded by microorganisms, whereas the more complex structures are more recalcitrant to degradation.
For example, Hexadecane is a very simple carbon chain (Fig.1 below) and usually disappears very quickly from the environment (1-7 days).

Hexadecane structure
Conversely, Phenanthrene has a much tougher structure (Fig.2 below) and can resist for a very long time (up to 1 year), travel with the sea currents and end up in our beaches.
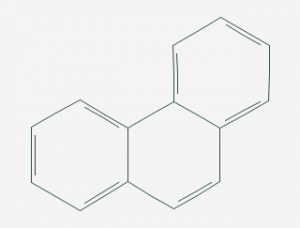
Phenanthrene
What are current best practices to limit the environmental pollution related to oil spills?
Let’s take the example of shallow releases of oil. When a given tanker is leaking or a land-based industry is releasing oil into the sea, there are several actions to be taken. The most important action is to contain the oil spill. For this, booms or barriers are deployed around the oil to concentrate it into a given spot, avoid its dispersal and facilitate subsequent phases. Once the oil is trapped, it can be burnt, or some dispersant can be used. The dispersants disaggregate the oil into tiny and numerous droplets making the oil accessible to natural bacteria. Another option is to use a skimmer which is a tool to suck the floating oil and collect it back.
However, some of these remedies, such as burning or using dispersants, add further pollution to the environment. In a simple way of thinking about it, we are fighting oil pollution with other types of chemical pollution. As such, this is not ideal and the scientific community is investigating eco-friendly alternatives.
Finally Grégoire, tell us a bit about these potential applications!
As a microbiologist, my goal is to make bacteria that are naturally available much more efficient in attacking the oil. There are two main ways to do this.
The first one is to add more nutrients into the system. The sea is not particularly rich in nutrients to promote dense bacteria population. However, we can use tools to deliver such nutrients and stimulate the activity and growth of bacterial communities. In turn, greater communities can degrade oil spills much faster.
The second option is to add directly bacteria that are able to attack the oil spill. Within laboratories, we can grow bacteria in controlled environments feeding them with the same oil that we want to degrade. As a consequence, we can select the ones that are more aggressive and let them reproduce in large quantities that are subsequently used during emergencies.
This sounds promising, but what are the real limitation of these techniques?
Well, if we choose to add nutrients we can never be 100% sure that the bacteria community that we are stimulating will be the ones that are able to degrade the oil.
On the other hand, if we choose to add laboratory-harvested bacteria directly we can incur into several issues. The first one is to add foreign communities into an ecosystem where they do not belong. This may sound trivial but it can actually trigger unforeseen consequences that we do not even know at the moment. In addition, the oil degradation typically undergoes several stages. Some bacteria will attack specific compounds and once they are weaker, another community will proceed to degrade the oil even further. However, these stages are reproducible in laboratory conditions, where our control limits the variability of the system (Temperature, Pressure, Oxygen, physical and chemical properties, etc). When we deal with an extremely complex system such as the seawater, the stages that are simulated in the lab, can be subjected to very different conditions and thus, the efficiency that we enhance in our research can be reduced or lost.
