Not long ago on the blog, we have talked about natural groundwater quality triggered by geogenic factors and related hazards such as a high concentration of heavy metals. Today’s topic concerns the anthropogenic input of heavy metals into the water and how to tackle its effects efficiently.
Industrial processes can lead to heavy metal-bearing wastewater, which is commonly treated by inefficient purification methods. Therefore, it is time to think about alternative methods for recovering the metals that would otherwise pollute the water and create a hazard for the environment. In addition, the obtainable metal resources make their recovery attractive from an economic point of view. However, efficient removal and recovery of toxic metals from industrial wastewater streams is a major challenge. To understand better the subject and the potentiality of specific water’s treatments, we interviewed two PhD candidates, Kai Tandon and Iphigenia Anagnostopoulos, both working in the group of Prof. Dr Soraya Heuss-Aßbichler at LMU Munich.

Iphigenia started her PhD in February 2018 at LMU Munich working on a pilot plant for the precipitation and recovery of metals from wastewaters at the industrial scale funded by the Bavarian State Ministry of the Environment and the Consumer Protection. Before she studied geoscience at the Munich Geozentrum with a focus on geochemistry and geomaterials. Her research interests are in the revision of the conventionally applied industrial methods from a mineralogical point of view.

Kai started his PhD at LMU Munich in 2017 working on new techniques to recover metals from salt-rich residual, e.g. fly ash of the municipal solid waste incinerator. Before that, he studied geoscience at the Munich Geozentrum focusing on applied mineralogy. His main research interests are to combine mineralogy, chemistry and physics to solve industrial technical issues, for example removing metals from solutions via chemical processes that take advantages of the detailed knowledge of the mineral structures.
What is industrial wastewater? What are the problems related to it?
Wastewater is a very broad term. The wastewater we are dealing with is generally a liquid solution rich in heavy metals from different industrial branches, for example, the electroplating industry. The toxic wastewater streams can contain one or more dissolved metal(s), like copper (Cu), cadmium (Cd), zinc (Zn), lead (Pb), but we have also worked with wastewater bearing noble metals such as silver (Ag) and gold (Au).
The main problem is that the conventional water purification methods produce hydroxide sludges that bind the metals within. Although the sludges are drained, they still contain huge amounts of water and in general, due to the high heavy metal content, they are environmentally hazardous. That is why the sludges are commonly disposed of in costly landfills and the metals, which are the most critical raw materials, disperse in the surroundings. In most cases, the metals bearing waste are deposited in such a way that we cannot recover it anymore.
What is the main hazard related to industrial wastewater?
In addition to the fact that we lose the metals as raw material, there are several hazards related to wastewater. One example is the accident that occurred at the Animas River near Durango, Colorado. 11000 m3 of contaminated wastewater was accidentally released from the nearby abandoned Gold King mine in August 2015 and flowed into the Animus and San Juan Rivers. The wastewater contained high concentrations of arsenic (As), Pb, and other heavy metals. Besides, in some industrial nations, most notably in China, loose environmental regulations and inadequate controls have led to serious pollution of natural water and soil because wastewater is introduced without purification in surface water. For example, the area around Lake Taihu, the third biggest lake in China, is one of the fastest-growing regions. Its water serves as a drink water source for about 10 million people, however, industrial and agricultural input pollute the lake with heavy metals. Some heavy metals are toxic for humans and animals as they can damage cells and organ functions. For example, Cd is already carcinogenic at low concentrations and if assumed regularly, it can cause the Itai – Itai disease responsible for bones’ softening and kidneys’ failure.
How are metals removed from wastewater? What are the main difficulties encountered?
There are various methods to treat metal-bearing wastewater; precipitation and ion exchange are the most common ones. Their main objective is usually to reduce the environmental impact of wastewater by purifying the solution. However, more and more research is going towards the removal and recycling of the metals contained in the solution. There are different ways to recover metals from wastewater – one has to find the right treatment for a single specific case. For example, we are developing a technique based on a concept we called ’SPOP: Specific Product Oriented Precipitation’ (Figure 1). This is based on the precipitation of components, in other words, specific metals and multi-element solutions, taking into account redox processes. We are looking into the reaction mechanisms or how the specific phases or products, such as metals and metal oxides, are formed. By individually optimizing the treatment conditions a product can be synthesized, that can be used for further processing. At the same time, after treatment, we obtain clean water that can be discharged into the municipal water system or directly in rivers depending on the final water conditions, for example, salinity content. Up to now, we have successfully developed a lab-scale treatment system for electroplating wastewater. The goal is now to up-scale the pilot plant for industrial-scale processes.
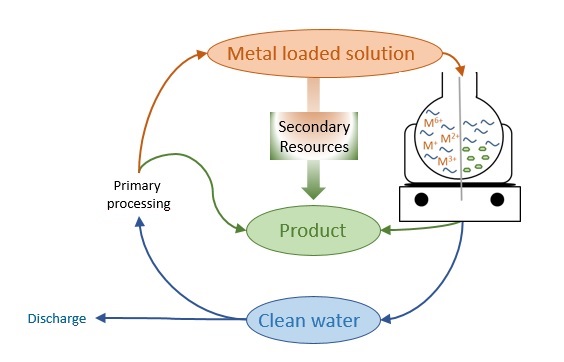
Figure 1 The SPOP concept sketched in a diagram. The metal loaded solution, i.e. the industrial wastewater, is processed in order to recover metals, oxides, etc., as products and clean water that can be discharged or reintroduced in the industrial cycle. Image credit: Iphigenia Anagnostopoulos and Kai Tandon.
Are the removed metals always recovered for secondary use?
It really depends on the treatment process that one uses. With the SPOP process, for example, we avoid the precipitation of the heavy metals as highly voluminous hydroxides. Instead, the heavy metals are precipitated as zero-valence phases (like Au0), oxides with a ferrite structure (like (Zn, Ni, Cu, Fe, Mn)2+Fe3+2O4) or delafossite mineral structures (like CuFeO2), and doped heavy metal oxides (like Cu2O, ZnO). The precipitated particles (Figure 2) are stable crystallites with a size of a few nanometres. In the worst case, the recovered particles can be used for metallurgical treatments. The goal is to make them attractive for industrial applications, for example, the doped ZnO is a compound valuable for semiconductor technology or piezoelectric devices.

Figure 2 Two examples of precipitation products recovered with the SPOP process. The black powder on the left is composed mainly of Jacobsite and it is recovered from an Mn-Fe rich solution. The white powder on the right is composed mainly of Gordaite and it is recovered from a Pb-Zn rich solution. Image credit: Iphigenia Anagnostopoulos and Kai Tandon.
What are the aims of current research on the subject?
Current research aims at economically recovering metals as anthropogenic resources. A first step in the right direction is to identify and classify anthropogenic resources. For this propose, the EU-network “MINEA – Mining the European Anthroposphere” recently published a document aiming to regulate globally the classification of anthropogenic resources. With such a standard, it is easier to identify resources and to show their potential for both public authorities and industry. We hope that our common efforts will contribute to raising awareness of avoiding geosphere abuses. There are ways to recover metals in an economic and ecological way: we only have to find them, optimize them and make them available. This process is clearly complex and represents a challenge that can only be tackled if academic, governmental and non-governmental regulatory institutions actively collaborate for a change in the use of secondary resources.




