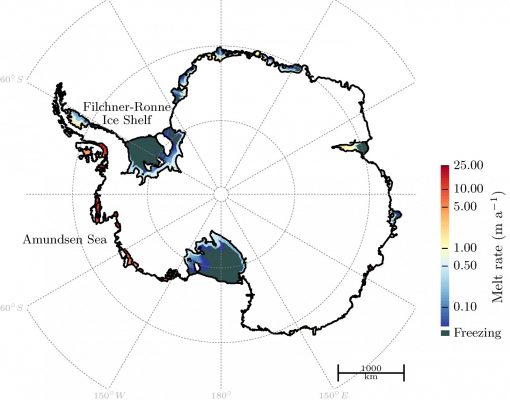
Between March and May 2007, a total of ~2,445 km2 (equivalent to over 17 football pitches) of ice mélange (a mixture of sea-ice types, icebergs and snow) and part of Voyeykov Ice Shelf in East Antarctica rapidly broke up. Observations of the timing and triggers of such events are relatively rare in East Antarctica, compared to ice shelves on the Antarctic Peninsula. Recent work highlights the impo ...[Read More]
It’s not you, it’s me(lange): ice shelf break-up triggered by mélange and sea-ice loss