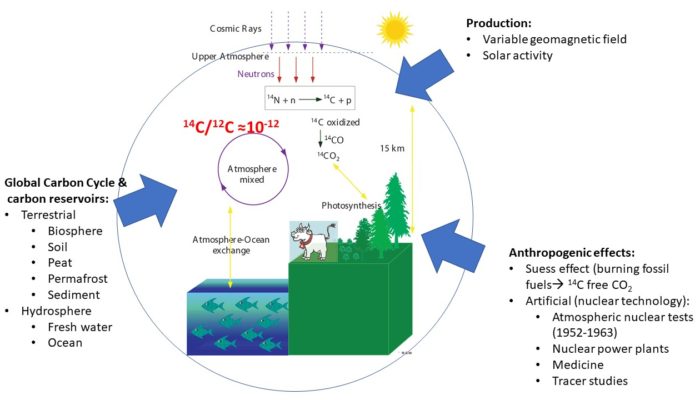
1950 CE is known as the year zero in the so-called ‘BP time scale.’ The BP stands for, “Before Present,” and has its roots in the development and conventions of radiocarbon dating [1]. The radioactive isotope of carbon (14C) with a half-life of 5700 years [2; 3] is a cosmogenic isotope produced in the atmosphere by secondary cosmic rays (SCR). Thermal neutrons, the SCR particle responsible for the production of 14C in the atmosphere, knocks one proton out of the 14N nucleus, transforming to form 14C as a product. Observations have shown that the atmospheric 14C content, often expressed as the ratio 14C/12C, fluctuates around 1.8 x 10-12.
There are different factors known to impact the 14C content on Earth (Figure 1). Variable strength of the geomagnetic field or solar activity results in a variable shielding of cosmic rays. A stronger geomagnetic field or more active Sun leads to lower production rates of 14C. Another source of the variability is changes within the global carbon cycle, i.e., the fluxes and the rate of exchange of carbon between various pools such as the ocean, soil, sediments, and the atmosphere. Anthropogenic perturbation also contributes heavily to the atmospheric 14C variability.
Anthropogenic perturbation of carbon into the atmosphere has had significant impact in the 14C record. In the 18th century, the advent of industrialization and burning fossil carbon increase atmospheric carbon dioxide (CO2) levels. This so-called Suess effect was first observed in tree rings as an ‘aging’ effect of the atmosphere due to the addition of ‘14C-free’ CO2 [5]. In the mid-20th century, this anthropogenic effect continued depletion of the natural 14C in the atmospehere. However, the advent of the nuclear era, marked by the post-WW2 thermonuclear tests, introduced a new source of 14C. The majority of the above-ground hydrogen bomb tests took place between 1952 and 1963, i.e., the year when the Partial Nuclear Test Ban Treaty was signed [6]. An excess of radionuclides released and spread around the globe as nuclear fallout is still detectable as a time marker of the mid-20th century [7]. Radiocarbon produced by the nuclear tests was soon detected in 14C measurements of contemporary organic matter and atmospheric CO2. Monitoring this unprecedented global rise of the atmospheric 14C content has become an essential part of radiocarbon research. One can only imagine the curiosity that drove the efforts to collect the atmospheric CO2 and measure its 14C content in years when the tests reached the apogee with the treaty looming on the horizon. Indeed, most detonations took place in the northern hemisphere and were recorded as the 1963 atmospheric peak, showing an offset of nearly 100% from the natural 14C levels [8]. The southern hemisphere observed a delay of ~1-2 years caused by the atmospheric circulation of 14C first injected into the northern stratosphere and then returned to the troposphere [9]. The efforts to monitor the atmospheric 14C levels, which began in the 1950s and 1960s, resulted in bomb 14C (B14C) curves compiled for each hemisphere and the atmospheric zones (Figure 2). The current set of compiled data is available via the CALIBomb web page and will be updated as soon as the new compilation is available (Hua et al., in preparation).
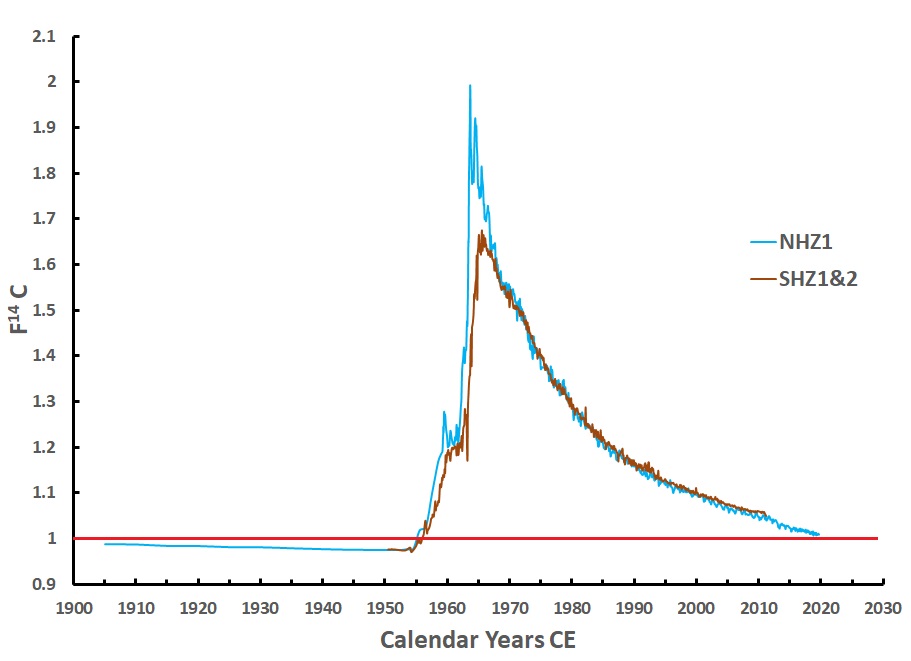
Figure 2. Northern Hemisphere B14C data sets (blue line) from Vermunt, Schauinsland, and Jungfraujoch [10; 11; 12] and a compilation of Southern Hemisphere data (brown line) [8]. The red line F14C (fraction modern carbon) =1 shows the average natural levels of atmospheric 14C. The first half of the 20th century shows depleted atmospheric 14C (F14C<1) due to fossil-fuel CO2 emissions.
In addition to the atmospheric data, 14C measurements of tree rings allow the extension of the atmospheric 14C record into various regions of the Earth. The presence of the B14C is detected when the measured 14C/12C is higher than the natural level of 1.8 x 10-12, i.e., measurement expressed as F14C shows values higher than 1 [13]. This clear signal can be detected in various carbon reservoirs such as the ocean or soils and can trace the global carbon cycle processes. Over decades extensive programs were dedicated to measuring B14C in the sea, which helped understand the movement of water masses. When built into annual bands of corals, the B14C is a valuable tool in reconstructing ocean upwelling history. Radiocarbon tracer has vast applications including biomedical studies, ecology, protection of wildlife, forensics, and detection of forgeries. Moreover, B14C dating can successfully be used in geochronology to date the top sedimentary deposits. The current investigations of sites in the ongoing formalization procedures of the Anthropocene focus on mid-20th-century deposits and use the B14C as one of the markers (see EGU CL blog).
However, the B14C is disappearing due to the ocean uptake and recently due to the Suess effect [14] (Figure 2; [12]). Consequently, radiocarbon measurements on samples formed in the future will return F14C<1 (Figure 2). The detection of so-called ‘modern’ carbon will not be possible without additional analysis such as stable isotopes and dating depositional sequences of samples.
This post was edited by the editorial board.
References [1] Libby, W.F., Anderson, E.C., and Arnold, J.R., (1949) Age Determination by Radiocarbon Content - World-Wide Assay of Natural Radiocarbon. Science 109, 227-228. [2] Kutschera, W., (2019) The Half-Life of 14C—Why Is It So Long? Radiocarbon 61, 1135-1142. [3] Kutschera, W., (2013) Applications of accelerator mass spectrometry. International Journal of Mass Spectrometry 349, 203-218. [4] Hajdas, I., (2008) The Radiocarbon dating method and its applications in Quaternary studies. Quaternary Science Journal - Eiszeitalter und Gegenwart 57, 2-24. [5] Suess, H.E., (1955) Radiocarbon Concentration in Modern Wood. Science 122, 415-417. [6] UNSCEAR, 2000 United Nations Scientific Committee on the Effects of Atomic Radiation UNSCEAR 2000 Report to the General Assembly, with Scientific Annexes volume I: Sources and Effects of Ionizing Radiation, https://www.unscear.org/docs/publications/2000/UNSCEAR_2000_Report_Vol.I.pdf, [7] Waters, C.N., Syvitski, J.P.M., Gałuszka, A., Hancock, G.J., Zalasiewicz, J., Cearreta, A., Grinevald, J., Jeandel, C., McNeill, J.R., Summerhayes, C., and Barnosky, A., (2015) Can nuclear weapons fallout mark the beginning of the Anthropocene Epoch? Bulletin of the Atomic Scientists 71, 46-57. [8] Hua, Q., Barbetti, M., and Rakowski, A.Z., (2013) Atmospheric Radiocarbon for the Period 1950-2010. Radiocarbon 55, 2059-2072. [9] Hua, Q., and Barbetti, M., (2004) Review of tropospheric bomb C-14 data for carbon cycle modeling and age calibration purposes. Radiocarbon 46 1273-1298. [10] Hammer, S., and Levin, I., (2017) Monthly mean atmospheric Δ14CO2 at Jungfraujoch and Schauinsland from 1986 to 2016. http://dx.doi.org/10.11588/data/10100 heiDATA: Heidelberg Research Data Repository [Distributor] V2 [Version]. [11] Levin, I., and Kromer, B., (2004) The tropospheric (CO2)-C-14 level in mid-latitudes of the Northern Hemisphere (1959-2003). Radiocarbon 46, 1261-1272. [12] Conen, F., Emmenegger, L., Leuenberger, M., Steger, D., and Steinbacher, M., 2020 14C Release, Jungfraujoch (10.0 m), 2016-01-04_2019-08-12, ICOS RI, 2020. ICOS ATC https://hdl.handle.net/11676/X-lXPKZlO4DWX7wncsLQ7akY. [13] Reimer, P.J., Brown, T.A., and Reimer, R.W., (2004) Discussion: Reporting and calibration of post-bomb C-14 data. Radiocarbon 46, 1299-1304. [14] Graven, H.D., (2015) Impact of fossil fuel emissions on atmospheric radiocarbon and various applications of radiocarbon over this century. P Natl Acad Sci USA 112, 9542-9545.



