
Post by Mark Cuthbert, Cardiff University, and Michael Rivett, GroundH20 plus Ltd; University of Strathclyde.
If you live in a cold climate, have you ever wondered where all the de-icing salt (or ‘grit’ as we call it in the UK) that gets spread on the roads in winter time ends up, aside from that accumulating salty grime that coats your car? As you might expect, most of the salt gets washed off the highways as the salt has the desired effect of melting the ice, or carried away by rain. This salty ‘runoff’ ends up in streams nearby via pipes which drain the highway. However, that is not the end of the story…
We studied a major highway intersection on the edge of UK’s second largest city, Birmingham (more than a million people), to see how much of the salt spread on the highway ends up in groundwater. Our interest stems from concerns of the national regulator to understand not only how much salt ends up in streams, but also the potential long-term build-up of salt in underlying groundwater resources that are pumped for public and private water supply. Although the origin of salt in streams and lakes is relatively well studied, the pathways by which salt moves from highways to groundwater are poorly understood and quantified.
The various ways salt could be getting in to the groundwater are shown in Figure 1 and a bit of detective work was required to find out what was going on.
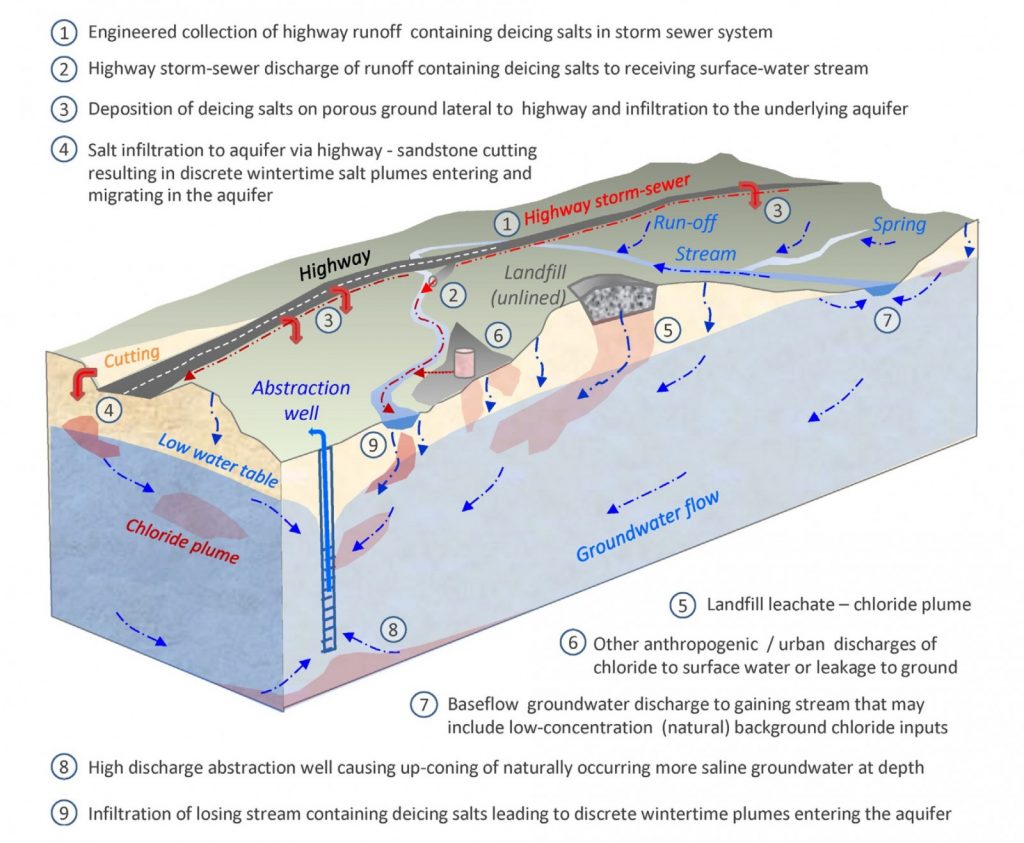
Figure 1*
This involved dressing in silly (and warm) clothes, and pulling our equipment around on a sledge, which was a lot of fun (see photos below).

The authors, Mark (left) and Mike (right), enjoying some urban winter fieldwork.
Our main field activities were fortuitously timed to include the severe winters of 2009-10 and 2012-13. We collected information about the amount of salt spread on the roads and measured the salt concentrations in the local streams, as well as in the shallow and deep groundwater wells in the area around the highway network.
7 km of motorway drained to the studied stream. Over the winter of 2012-13 we estimated that around 510 tonnes of de-icing salt was applied to the highway and major road network across the catchment. Most of that washed off the road via drains when it next rained or as the snow and ice melted, and ended up in the stream which flows under the highways. Some of it ended up on the roadside verges – this was not quantified, but would likewise eventually leach into the underlying groundwater over time.
We estimated about 12% of the de-icing salt, that’s around 63 tonnes over the 2012-13 winter, leaked through the bottom of the stream channel into the groundwater in the sandstone aquifer beneath, and may pose a risk to groundwater supplies in the area (see “9” in Figure 1, and results in Figure 2). While increases in groundwater chloride concentrations observed to date have been modest, and fortunately remain far below the drinking water standards, the steady year-upon-year build-up of salt in groundwater remains a concern. This is especially the case as the UK’s de-icing salt applications reached record levels in the recent severe winters and add to a potential de-icing salt legacy in our aquifers that has accumulated now over some 50 plus years.
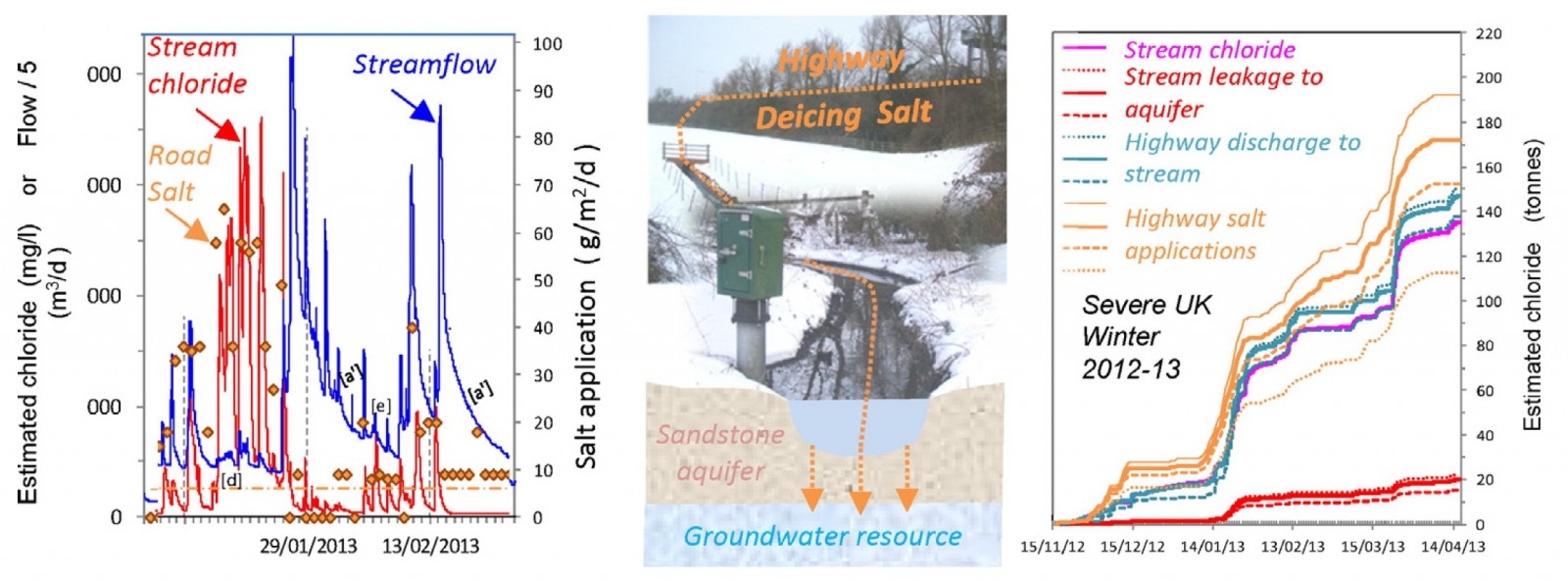
Figure 2*
So how concerned should we be that so much salt is flowing down these streams and into the groundwater around our highways? There isn’t yet a simple answer to that but, with a global trend towards increasing urbanisation, this is an important area of ongoing and future research.
Further details of our published research to date on the Birmingham case study may be found at:
Rivett, M.O., Cuthbert, M.O., Gamble R., Connon, L.E., Pearson, A., Shepley, M.G., Davis, J., 2016. Highway deicing salt dynamic runoff to surface water and subsequent infiltration to groundwater during severe UK winters. Science of the Total Environment 565, 324-338. http://dx.doi.org/10.1016/j.scitotenv.2016.04.095 (*figures reproduced with permission)




Andrew Dawson
Highway agencies are very well aware of the problem – indeed, they’d be very happy to spread less salt so as to save cash! But they are under huge pressure from road users to keep roads clear of ice. There are more benign de-icing chemicals, but they cost a lot more (of course). In many circumstances the use of brine spray as temperatures fall maybe the best solution (no pun intended!). This forms a thin coating on the road surface and prevents ice forming. Much less salt is needed than when adding granular salt after ice formation – and adding brine after salt formation isn’t nearly as good (the brine runs off). The tricky thing for a highway agency is to get the brine applied at the right time – not too early otherwise it will be lost, not too late or it wont work well. An implication would be a large fleet of applicators on call to spray … another cost! In the UK with regular cycles of temperature above and below zero in winter brine could work, but in countries where there is snow and ice build up and few winter transitions to >0deg, granular salt remains one of the few practical options. The report at http://www.dot.state.mn.us/research/TS/2014/201443.pdf may be of interest.
Andrew. (Nottingham Transportation Engineering Centre)