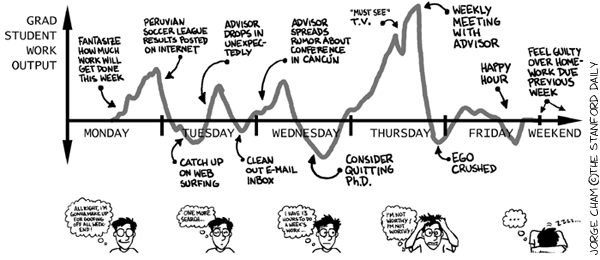
Today we have a new guest post written by current PhD candidate and Antarctic researcher on her very fascinating field work. Actually, she wrote this post while at McMurdo station. Hilary and I have known each other since our time at Queens University in Kingston, when she was one of my TA’s and was doing her masters. For more info about her work see the bio at the end of the post at check out her own excellent blog.
In Antarctica, there is a small swath of land hidden by the Transantarctic Mountains that is too dry and sheltered to be overridden by the ice sheets that cover over 99% of the continent. In these barren valleys, life is at the edge of existence and sustained by pulses of meltwater that form when summer temperatures finally break the freezing point. The only refuges of perennial water in this habitat are the large lakes that occupy the topographic depressions in the valley bottoms. The lakes themselves are hidden beneath permanent ice covers of 4 m, but reach depths of 20 to 75 m, and temperatures of -13 degC to +25 degC. As a colleague remarked, “This system of valleys is one of the coldest and driest places in the world and has more in common with Mars than it does with your backyard”.

Don Jon Pond (the saltiest body of water on Earth) in Wright Valley, Antarctica
The McMurdo Dry Valleys is also one of the only places in the world with year-round lake ice, which is partly why I spend a few months of the year hidden beneath a giant red parka. Imagine studying the atmosphere without solid ground. Where would we build telescopes, satellite dishes, or research stations? In oceanography and limnology, this is a fundamental roadblock in the collection of long-term data, and is amplified in remote and deep environments where ships or divers can be logistically impossible to send.

Downloading datalogger at Lake Fryxell, Antarctica
Luckily for polar researchers (and everyone else *), the solidification of water into ice happens at relatively warm temperatures (0 degC for freshwater, -1.9 degC for ocean water) and floats, thereby providing a frozen platform to access the hidden ecosystem beneath. In most temperate environments, this advantage is limited to a few winter months, and in the shoulder seasons, ice is viewed as a destructive force capable of destroying or dragging around all but the sturdiest of instrumentation. The result is most high-resolution lake data is acquired from spring to fall before buoys are pulled for the winter. Ice is both a boon and a barrier for limnology.

Lake Bonney, Antarctica. Blood Falls can be seen at the edge of Taylor Glacier in the lower right hand corner.
The permanent ice covers in the Dry Valleys allow us to moor instrumentation beneath the ice cover year round, which is a rarity in limnology. Below 4 m of ice, we record physical parameters, such as ice thickness, underwater radiation, ice ablation, and lake level. Our research is primarily focused in Taylor Valley, which has been an US National Science Foundation LTER (Long Term Ecological Research) site for the past twenty years. Three large closed-basin lakes span a range of physical conditions, and represent some of the saltiest and coldest bodies of water on Earth. Because the lakes harbor liquid water year-round, the lakes may be the microbial Amazon of the Dry Valleys; even though the ecosystem is made up of a simple trophic structure. This simplicity allows biological processes and interactions to be more easily studied than in more biologically complex habitats.

Surface dataloggers at Lake Hoare, Antarctica
This long-term data allows us to track the habitability of lakes, and general hydrology of the watershed. For instance, the last decade has seen a tremendous rise in lake levels, and therefore a positive water balance in the Valleys. This has come without a concomitant increase in temperature, and researchers are currently investigating the trigger for meltwater production in this water-starved environment.

In our goal of year-round monitoring, one major hurdle is that biologic sampling is only conducted during the summer, when the temperatures are reasonable enough for personnel to be in the field. Therefore, any assumptions of microbial activity during the polar winter have been extrapolated from data procured mainly from Oct to Jan (one very cold season stretched until April).
This field season, our goal was to fill in the missing months, and for the first time understand ecosystem functioning during a period of total darkness; a subject extremely valuable to those studying the habitability of environments outside our planet. Instead of over-wintering in Antarctica (we’re not that crazy), we moored three large automated instruments in Lake Bonney: a water sampler, a phytoplankton sampler, and a profiling CTD equipped with a fluorometer and CO2, dissolved oxygen, and PAR sensors. These instruments will be collecting data and samples until our return in Nov. 2014.

Deployment of an automated phytoplankton sampler in Lake Bonney, Antarctica.
Pictured: Luke Winslow (University of Wisconsin, Madison), Kyle Cronin and Dr. Peter Doran (University of Illinois, Chicago)
This winter it will be 40 years since the New Zealand program’s last overwinter campaign in Wright Valley. While they braved complete darkness and colder temperatures than most of us have ever experienced in the pursuit of meteorological measurements, I will be nestled warmly in Chicago knowing that somewhere far away a CTD will be capturing the first winter data from one of the most unique lakes on the planet.
As otherworldly as Antarctica may seem, the life that exists in this frozen corner of the Earth demonstrates the incredible adaptation of organisms to surrounding environments, and is likely the closet planetary analogue to any life that may exist on other icy planets in our solar system. Perhaps one day in the future, some young scientist will be making the same comments about their research beneath the icy shell of Europa.
* – If ice was denser than water (like the solid form of most liquids) the ocean would freeze from the bottom up, drastically changing ocean circulation and climate.
I am a PhD candidate at the University of Illinois at Chicago, working with Dr. Peter Doran in the Department of Earth and Environmental Sciences. My current research focus is on Antarctic limnology, with the overarching hypothesis that small variations in climatic conditions can result in extreme hydrologic shifts. I am actively involved in three Antarctic projects: one which examines the hydrology and microbiology of a unique lake with a 27+ m ice cover, a second which uses geophysical techniques to map subsurface brines beneath lakes, and a third which focuses on long term limnological changes as part of the McMurdo Dry Valleys Long Term Ecological Research (LTER) program.

One of my responsibilities is to maintain long-term data sets associated with the physical properties of the McMurdo LTER lakes. This includes field-based implementation of lake stations, upkeep of instrumentation, data compilation and management, and ultimately, analysis of the data sets. I regularly employ analytical tools, such as R, Matlab, and ArcGIS, to both to post-process data and explore spatial imagery.
For more information, feel free to visit: https://sites.google.com/site/hilarydugan/
Or check out my field blog at: http://b511m.wordpress.com/