Ok, so I took some license with the title. This isn’t really a curious case and neither Krypton-81 nor ATTA are actually people. In fact, Krypton-81 (81Kr) is a radioisotope of the noble gas krypton and ATTA, which stands for atom trap trace analysis, is the revolutionary technique that has made its analysis possible. I recently heard about developments with ATTA at the IAEA Isotope Hydrology Symposium and have been doing some reading about the method and its revolutionary application to the dating of both young and ancient groundwater.
81Kr has long been a bit of a dangling carrot for groundwater dating people like myself. 81Kr is a long lived radioisotope of Kr (half-life: 229,000 years) that is produced by cosmic ray interaction in the atmosphere with other krypton isotopes. This production results in about 5 atoms of 81Kr for every 10^13 atoms of the other Kr isotopes. This 81Kr then settles to the earth surface and is incorporated into groundwater recharge and can then used to date groundwater from 150 thousand to 1.5 million years old. The way this works is that once water reaches the water table no new krypton is added and the clock starts ticking as the 81Kr decays away. In order to use this method we assume that the initial concentration in the recharge is in equilibrium with the concentration of 81Kr in the atmosphere, which is well mixed. ATTA then measures the amount of 81Kr that is left in the water sample compared to the other Kr isotopes and an age can be calculated from the difference between this ratio and the intial ratio.
ATTA can also be used for the short-lived isotope krypton-85 (half-life: 10.8 years). 85Kr is produced by fission in nuclear reactors and is released during nuclear fuel reprocessing. The short half life of 85Kr makes it useful for dating recently recharged groundwater from 1 to 40 years old.
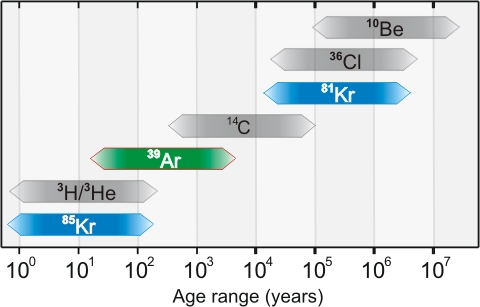
Figure 2. Dating ranges of 85Kr, 39Ar, 81Kr and other established radioisotope tracers. (Source). Used with permission.
The reason krypton is such a useful tracer for groundwater dating is that as a noble gas the interaction of Kr with soils, rocks and the biosphere is minimal whereas other tracers such as 36Cl, 14C or 3H are often subject to retardation during transport or inputs from multiple sources which makes extensive corrections necessary or renders them completely unusable for dating. Furthermore, very few reliable tracers exist in the range that Kr isotopes cover making them extremely useful. One isotope that I haven’t mentioned as much is argon-39, which can be used to date water from 50-1000 years old, is also a noble gas, and can also be measured with ATTA.
Measurements of krypton can also be used for dating of ancient ice cores as well. Atmospheric gases including Kr are trapped in air bubbles in the ice and therefore, using the same method as groundwater dating, an absolute age for an ice core can be obtained. There are several other applications for Kr dating as well such as dating of deep crustal fluids and brines.
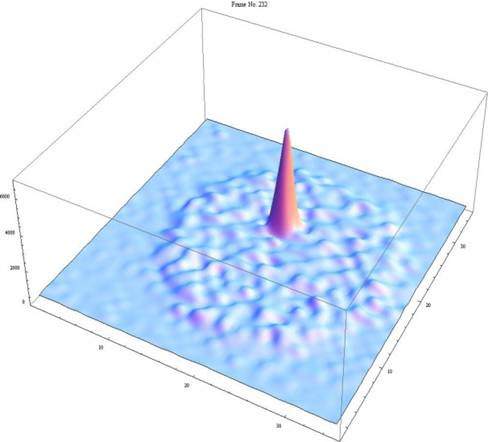
The development of atom trap trace analysis was first reported in Science in 1999 and since then has undergone several substantial improvements primarily aimed at reducing the required sample size required for an analysis of Kr. ATTA (Figure 4) works by trapping Kr atoms with a laser which causes a slight and temporary change in their atomic structure which lasts for about 40 seconds. During this period the Kr atoms in the laser beam are focussed and slowed and then trapped in an MOT (magneto optical trap) where they are held in place for an average of 1.8 seconds. Once the Kr atom is in the MOT it fluoresces as it returns to its original state. This fluorescence is detected by a camera which is sensitive enough to detect the emission from a single atom (Figure 5)!
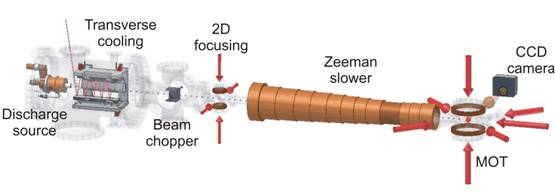
Figure 4. Schematic layout of the ATTA-3 apparatus. (Source). Used with permission.
One of the key features of ATTA is that this laser induced fluorescence within the MOT occurs uniquely for every isotope as the laser frequency is tuned specifically! This means that only atoms of of the desired Kr isotope are trapped. Furthermore, this means that ATTA is completely immune to interference from other elements, isotopes, isobars, or molecules. In essence nothing can confuse the detection of the 81Kr atom once it fluoresces and therefore there is no background of spurious detections that need to be corrected for. Among low-level analytical techniques this is unique and a really big deal! As a user of AMS, which is another low level analytical method that does suffer from these issues, this is statement is an eye-catcher.
Since its invention ATTA has evolved considerably. We are now on the 3rd iteration of ATTA and significant improvements have been made that make ATTA much more practical for routine use. Specifically, the amount of sample required for an analysis has been reduced drastically. The first version of ATTA could only be used for atmospheric measurements as the quantity of Kr needed was too large to be extracted from water. ATTA-2 required ~1000 kg of water to extract 50uL of Kr gas. Now, ATTA-3 only requires 5-10uL of Kr which can be obtained from only 100-200 kg of water or 40-80 kg of ice. This advancement means that ATTA is now usable for groundwater dating applications never before possible. This has been demonstrated by the use of ATTA to date groundwater in Egypt to around 500,000 years old and even older water in Brazil up to 800,000 years. Other dating methods have confirmed that ATTA measurements are accurate.
Now that the sample sizes required for an 81Kr or 85Kr analysis have been lowered so dramatically the method is even more useful to the geoscience community. One of the messages from Dr. Lu’s talk at the IAEA meeting was that this technique is open for business and the geoscience community is strongly encouraged to reach out for collaboration and discussion. Furthermore, it may also be possible to use ATTA to measure argon-39, calcium-41 and potentially lead-205, strontium-90 and cesium-137,135 at extremely low levels.
Note: During the writing of this blog I corresponded with Dr. Z-T Lu, one of the creators of ATTA. I would like to thank him for allowing me to use his personal photos in this post. Dr. Lu is now establishing a radiokrypton dating centre at the University of Science and Technology of China.
References
Lu Z-T, Schlosser P, Smethie WM, Sturchio NC, Fischer TP, Kennedy BM, et al. Tracer applications of noble gas radionuclides in the geosciences. Earth-Science Rev. 2014;138:196–214.
Chen CY. Ultrasensitive Isotope Trace Analyses with a Magneto-Optical Trap. Science (80-). 1999;286(5442):1139–41.
Du X, Purtschert R, Bailey K, Lehmann BE, Lorenzo R, Lu Z-T, et al. A New Method of Measuring 81Kr and 85Kr Abundances in Environmental Samples. Geophys Res Lett. 2003;30(20):2068. Available from: http://arxiv.org/abs/physics/0311118
Aggarwal PK, Matsumoto T, Sturchio NC, Chang HK, Gastmans D, Araguas-Araguas LJ, et al. Continental degassing of 4He by surficial discharge of deep groundwater. Nat Geosci. 2014;8.
Lu Z-T. Atom Trap, Krypton-81, and Saharan Water. Nucl Phys News. 2008;18(2):24–7.