Phosphate use for fertilisers, essential in modern agriculture, is hitting an all time high while resources are being heavily depleted. Flo discusses the background, numbers, geopolitics and potential solutions behind the issue of ‘the end of phosphorus’.
The Issue

Modern agriculture has developed in-line with the availability of high quality phosphate-rock fertilisers. Source – João Felipe C.S, Wikimedia Commons.
The dilemma over diminishing natural resources is a topic of our times with the daily bulletins filled with reports related to resource shortages. These mainly focus around water, energy and food which are imperative for human survival. Whilst energy and water are often debated in the media and political chamber, an area that gets much less attention is agriculture, and in particular diminishing phosphate resources used for industrial fertiliser. Modern agriculture, particularly in developed countries has used mined phosphate for fertilisers for decades but this finite resource is being depleted at an alarming rate.
A combination of growing population, aspirational lifestyles and the demand for phosphate-intensive meat and crops has caused the rapid reduction of phosphate rock resources. In the past, prior to the advent of phosphate mining, additional phosphate for farming and agriculture was sourced from manures

Prior to use of phosphate rock, it was replenished through the use of manure. Source – Malene Thysson, Wikimedia Commons.
and organic waste, but as agriculture intensified, the hunt for easier, more accessible phosphate began. From the mid-20th century onwards, the use of rock phosphate was used as a high quality, easily accessible sources of phosphorus which gave rise to the modern fertiliser industry as we see it today. Farmers in rich countries such as Europe and North America became hooked on the cheap and easy phosphorus which readjusted agriculture practices and set phosphate demand through the roof.
Background
Phosphorus (P) is a non-metallic element which is almost always present in a maximally oxidised state (PO43-) as inorganic phosphate rocks due to its reactivity. Elemental phosphorus can exist as red and white (known for its use in weapons and artillery) phosphorus but almost never found as a free element in nature.
It is one of the building blocks of life and life simply wouldn’t exist without it. It is a key component of DNA, RNA, ATP and phospholipids and is essential to cell development, reproduction and in animals, bone development. The use of phosphorus compounds in fertilisers is due to the need to replace the phosphorus that plants remove from the soil.There is no substitute for this element. Supplies are limited and much is currently wasted, creating concerns about future supplies in the EU and worldwide.
Peak Phosphorus?
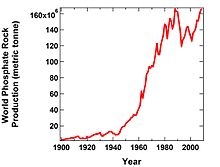
A graph of world phosphate rock production vs. year from 1900-2009 obtained from the U.S. Geological Survey. Source – Thomas D. Kelly and Grecia R. Matos, Wikimedia Commons.
Recently there has been a proliferation of articles and discussion over the potential for ‘peak phosphorus’ in the next 20-30 years. World production recently peaked at <160 million metric tonnes (mmt) in 2008. Whilst the majority of people agree that phosphorus is a resource that is of concern, not everyone agrees with the peak phosphorus hypothesis or its potential timing. Proponents of the argument include this group of academics who published a paper entitled ‘The story of phosphorus: Global food security and food for thought’ and Jeremy Grantham, co-founder of the investment firm Grantham, Mayo, Van Otterloo, who recently wrote a piece in Nature. On mined-phosphate fertilisers, Jeremy Grantham stated that ‘There seems to be only one conclusion: their use must be drastically reduced in the next 20–40 years or we will begin to starve’. Much of the peak phosphorus argument comes from a widely produced diagram from a 2009 paper in Global Environmental Change depicting peak phosphorus to be around 2030 followed by production declining at an accelerating rate. Detractors to this theory say that the markets are likely to adjust to this problem and cause the price to rise thus forcing a reduction in use and a push for technology to advance to find new sources or recycle current phosphate use. The International Fertiliser Development Center (IFDC), through extensive data gathering state that there is “no indication that a “peak phosphorus” event will occur” in the next 20-25 years.
Resources and Geopolitics

Phosphate mine near Flaming Gorge, Utah. The large size of phosphate mines is dictated by the dispersed nature of phosphate in the rock. Source – Jason Parker-Burlingham, Wikimedia Commons.
Phosphate rock is typically mined at high volume due to its dispersed nature in the rock. Phosphate, in the mineral form of apatite in phosphate rock, is not bioavailable to plants and must be processed to convert it to a plant-available form. The concentrate is used to produce phosphoric acid which is then used in fertiliser products. Phosphate rock can come in either a sedimentary or igneous form, with sedimentary making up >80% of total global production.
Topographic map of Western Sahara. Western Sahara is disputed territory but is currently controlled by Morocco and therefore the Moroccan Royal Family. Source – Sadalmelik, Wikimedia Commons
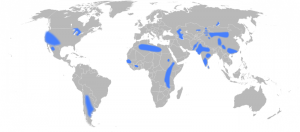
In addition to the concern over the amount of resources and rate of use, also of concern is the location of much of the world’s supplies. Europe in particular has scant phosphate resources with a small amount in Finland. According to the IFDC report from 2010, 72.1% of the world’s phosphate rock production was accounted for by China ( 31.5%), U.S.A (18.7%), Morocco and Western Sahara (15.5%) and Russia (6.4%). However a significant proportion of the world’s high-grade supplies are located in the disputed territory of Western Sahara in North-West Africa, currently controlled by Morocco. This has been termed by Jeremy Grantham as ‘the most important quasi-monopoloy in economic history’.
| Country | Mine Production 2007 |
Mine Production 2008 |
Reserves | Reserve Base (estimated) |
| China | 45,700 | 50,000 | 4,100,000 | 10,000,000 |
| Morocco and Western Sahara | 27,000 | 28,000 | 5,700,000 | 21,000,000 |
| Russia | 11,000 | 11,000 | 200,000 | 1,000,000 |
| United States | 29,700 | 30,900 | 1,200,000 | 3,400,000 |
| World Total (rounded) | 156,000 | 167,000 | 15,000,000 | 47,000,000 |
Table adapted from USGS Mineral Commodity Summaries January 2009. Data is presented in thousand metric tonnes.
Phosphate rock resources in Western Sahara are extremely large and still incompletely explored and therefore it is not understood if the rock is producible at current prices and costs as there is little to no data. The IFDC estimates global resources of 290,000 mmt but if Morocco and Western Sahara resources are included (340,000 mmt) it may increase to 470,000 mmt, as seen in the above table.
Environmental Impacts

Eutrophication in a pond in Lille, France. Eutrophication is caused by the enrichment of an ecosystem with chemical nutrients such as phosphorus. Source – F. lamiot, Wikimedia Commons.
The use and mining of phosphorus also carries risks. In agricultural use not all of the phosphorus is absorbed by crops which results in leaching into the water. This causes the much discussed eutrophication effect causing algal blooms. Phosphorus mining is also difficult environmentally as it generates a large amount of the waste product phosphogypsum which contains both toxic heavy metals and low levels of radiation. This is very difficult to dispose of and often results in mounds of unprocessed waste material.
As lower-cost phosphate resources will be mined out, mining companies will utilise lower grade ores which will incur the use of more energy and water and will cause the price to go up. Availability of water is of high importance to mining operations and indeed this can dictate the feasibility of phosphate extraction, areas of low water availability may completely restrict development of the mine. Another way in which water, energy and food are interconnected.
What next?

Wastewater discharge pipe – New studies show useable phosphorus can be recovered from wastewater. Source – Department of Agriculture, Wikimedia Commons.
Regardless of the proximity of ‘the end of phosphorus’ it is very much a finite resource and thus development towards more effective use and recycling needs to take place. Recently, the Environment section of the EC launched a consultation into how to use phosphorus in a more sustainable way, following on from a conference held in March on Sustainable Phosphorus. They have also posted a series of informative videos that can be found here. Much work has been done on the ways in which we can curb our phosphate use or recycle it more effectively. More must be done to monitor and reduce phosphate use as well as recycle wasted phosphate. A few of the potential solutions are listed below.
Phosphate reduction
- Changes in people’s daily diet away from phosphorus-intensive foodstuffs such as meat.
- Since much of phosphorus is lost from the food cycle through waste, a reduction of food waste and its reuse in composts etc could reduce demand.
- Current agricultural practices result in a very high use of fertilisers. A switch to techniques and practices that conserve more soil nutrients would go some way to reduce phosphorus waste. This includes organic agriculture and use of permaculture (sustainable and self-sufficient agricultural practices).
- Genetic engineering could produce plants that can flourish with much lower phosphorus use.
Recycling
- We can recover useable phosphorus from waste streams including urban sewage, since current systems already remove phosphorus from sewage to preserve water quality. Wastewater carries a lot of struvite, a mixture of ammonium, magnesium and phosphate which builds up in the pipework.
- A team of canadian researchers believe struvite can be turned into environmental friendly fertiliser, as discussed in this national geographic article. Together with the local government they have set up a lab next to a waste water treatment plant. This process works by altering the pH and allows the wastewater chemical to bond together into pellets through a turbulence process. Currently a working prototype can turn out several tons of pellets a month. Since 2010, the technology has been incorporated into 5 waste water facilities in North America. Whilst there are some cost issues to address, there is relatively little further work required to reproduce this technology on a wide scale. Higher phosphate prices would push wastewater recovery to be economic.