The ‘rare earth elements’ (REEs) are a series of similar elements that are rarely encountered on the Earth’s surface. They’re not even allowed to sit in the proper periodic table, banished to a small row floating below with only the ‘actinide series’ (the what?!) for company. REEs have always been the lonely kid in the playground, but now our mum is making us invite them to our birthday party. Like it or not, REEs are of growing economic, political and environmental importance.
Periodic table of the elements, with rare earth metals (including scandium and yttrium) circled. Image from tnahistoryoftechnology.com
Geochemists, such as myself, try to extract REEs that have been trapped and preserved inside sedimentary rocks and use them to reconstruct ancient environments. When the normalised concentration of each REE is plotted in a series, a distinctive distribution pattern emerges for sediments that formed in seawater (pictured below), showing enrichment in heavier rare earths. Most REEs behave in a similar and predictable way, but some of the elements within the group have unique properties. Cerium, for example, is different to most other REEs because it can exist in two different oxidation states. Cerium will behave the same as the other REEs when in anoxic water, such as the Black Sea or Precambrian oceans, but in oxygen rich conditions cerium is preferentially partitioned into iron oxide phases, leaving seawater depleted in cerium.
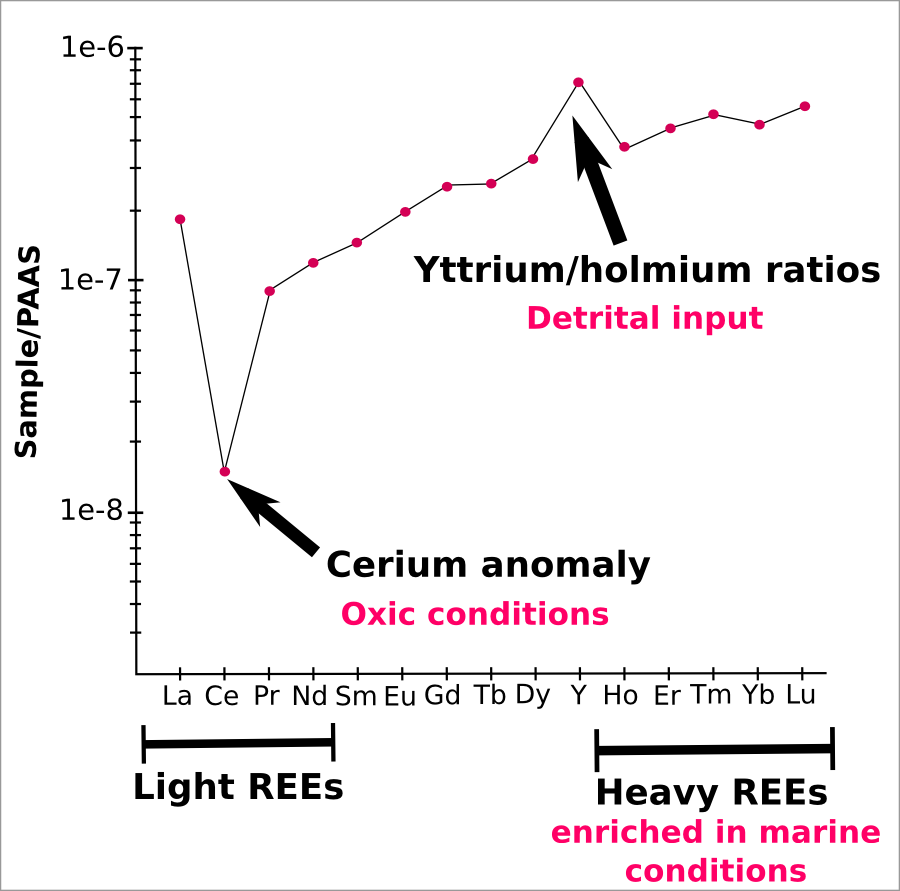
Typical open marine REE pattern. Key features are highlighted in black, and their main geochemical use in pink. Y-axis is a log scale, and values are normalised to ‘PAAS’ (a shale). (c) Rosalie Tostevin
As well as telling geochemists about the redox state of the oceans, cerium has economic value as a chemical oxidising agent. Other REEs are used in products such as lasers and permanent magnets. REEs are essential components in some military equipment and in many newly emerging renewable technologies.
Rare earth element deposits are indeed rare, and their scarcity makes them lucrative. Almost 50% of global REE reserves (55m tonnes) lie in China, and China unsurprisingly dominates the world market. They have been able to produce rare earths at lower costs due to favourable deposits, weak environmental regulations and cheap labour. China announced they would be dropping their exports by 70% in the second half of 2010, resulting in a global spike in REE prices.

Rare earth oxide powders. Clockwise from top center: praseodymium, cerium, lanthanum, neodymium, samarium, and gadolinium. Image from US department of Agriculture
Because of the rising price and increasing scarcity of REE deposits, people are starting to gaze out to sea. Speculation surrounding the possibility of mining metal rich seafloor nodules has been going on for decades. These nodules grow slowly, so they adsorb high concentrations of metals from seawater. Seafloor deposits have a high ratio of the heavy, more valuable, rare earths to light rare earths compared to land deposits. This resource has not been widely exploited before now because mineral deposits available on land are cheaper and easier to access. Recently, a British company secured a UN permit to explore a large area of the Pacific seafloor.
Geologists know that the seafloor is home to a wide range of complex processes: redox gradients, bioturbation, microbial communities, mineral precipitation etc. Can we really be sure of the consequences if significant volumes of metal rich nodules are removed, noisily churning up the sediment and destroying benthic habitats?
On the seabed the international boundaries are not clearly marked, but we have a shared interest in protecting our oceans. Poor decision making in one part of the ocean will spread around the world at about the same rate as ocean currents move. Just as debris from the Japanese tsunami is beginning to haunt American shorelines, we could see the effects of poor environmental legislation washing up in every nation.
The technology to explore the seabed is already developed, some of it borrowed from the oil industry, and is waiting for the go-ahead to start mining. At the current rate of progression, rigorous scientific monitoring of the environmental consequences will not be able to keep up.
Aside from environmental concerns, seafloor mining will face some of the same political and economic problems that the resource extraction industry faces on land. Who will keep the profits? Where are companies obliged to pay taxes? Will the resources be managed and traded fairly and responsibly? The reserves are concentrated in the Pacific Ocean, on the seafloor surrounding both Hawaii and French Polynesia.

Satellite image of the Iles Gambier, French Polynesia Image from NASA
French Polynesia, a group of isolated volcanic islands in the southwest Pacific, has built its economy on pearls and tourism. Their history, their economy and even their transport system is intimately linked to the ocean surrounding them. Unemployment sits high at 13% and twenty percent of the population live below the poverty line – they are dependent on financial help from France. REE mining could offer a big opportunity for French Polynesia, but right now it is unclear exactly how the profits from ‘the modern gold rush’ will benefit the islanders.
Recommended reading
A detailed report on seabed mining from the University of Southampton.
The BBC write about licenses being distributed to british mining companies.

Giuseppe Di Capua
Thank you Rosalie for this really interesting article. I appreciate your question: “Will the resources be managed and traded fairly and responsibly?”. This is a big problem from a geoethical point of view: is it possible a sustainable development? The answer seems easy, but how to do it is really difficult…
Kind regards,
Giuseppe Di Capua
(International Association for Promoting Geoethics: http://www.iapg.geoethics.org)
Mohammad Omer Faruk Khan
Really a very engrossing article.
Jesse
About the seawater chart. Is that ~0.0000001 RE per shale on the y-axis? What are you dealing with in terms of ppm’s of RE in the sediment? If we’re looking at sedimentary rock formed in the ocean then shouldn’t we see more cerium in the sediment under “oxic” conditions? The depletion of cerium looks more like what one might expect in the actual seawater.
Rosalie Tostevin
Hi Jesse,
The diagram shows a typical seawater REY distribution, so you are right – this is the water column. The y axis has been normalised to shale (PAAS).
We think that the seawater REY distribution is transferred faithfully into carbonates. In oxic conditions, cerium would be oxidised to +4 and precipitated within iron and manganese oxides, leaving both seawater and carbonates depleted in cerium.
Hope that answers your questions?
Rosalie