This was originally posted on James Lewis’ personal blog at:
http://marsblogger.wordpress.com/2014/12/08/minerals-and-the-search-for-life-on-mars/
(Re-posted with permission)
Understanding if life could ever have existed on Mars is one of the most challenging scientific questions facing us in the 21st Century. We know that the Martian surface at present is an arid environment bombarded with ultraviolet radiation, so the chance of finding living organisms existing there today is extremely unlikely. However, Mars has not always been this way, its history is divided into three distinct geological periods; the Amazonian, Hesperian, and the Noachian. The oldest of these, the Noachian, is likely to have been a significantly more promising time for life to potentially evolve as liquid water persisted on or near the surface long enough to carve valleys into the Martian surface and leave behind distinctive rock units. For example, in Gale Crater Curiosity Rover discovered minerals that indicated the presence of a freshwater lake at the time of their formation billions of years ago, an environment favourable to life or at least life as we understand it on Earth.
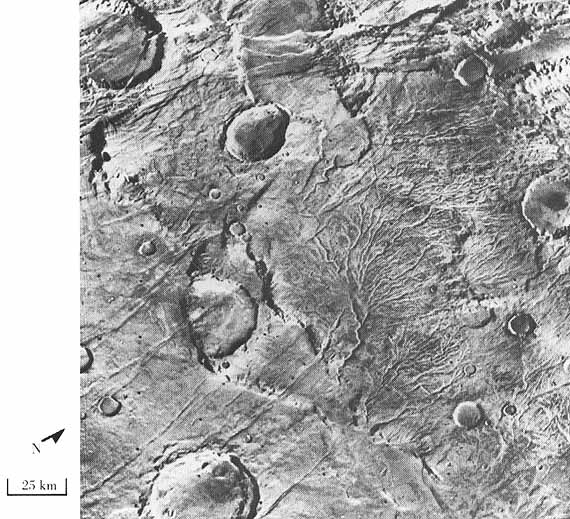
Dendritic channels in the ancient Southern Highlands of Mars. From NASA history
Though life could potentially exist deep underground on present day Mars the most realistic aim, with our current technological abilities, is to examine ancient Martian rocks for pieces of evidence that life could have left behind. Any life that may have existed on ancient Mars would likely be microscopic so looking for fossil evidence is extremely challenging and many microscopic features that look like fossils can often be formed by non-life processes. The most unambiguous result would be to find carbon compounds on Mars that we know form through biological means. Life leaves behind many distinct carbon compounds but not all carbon compounds, which are referred to as organic molecules, are evidence of life. Many form by non-biological processes, such as reactions between carbon dioxide, water and rocks at high temperature or the accumulation of dust and grains in the early Solar System. We know that cosmic dust, asteroids and comets are packed with complex organic molecules. Even if life never existed on Mars we would expect to see the organic compounds added to the planet by the regular impacts from these bodies.
There have been several missions sent to Mars for which searching for organic compounds was a high priority (the Viking Landers and Curiosity Rover). However, none of these have made a conclusive detection of either biological or non-biological indigenous carbon compounds. All that has been detected so far are simple carbon and chlorine compounds that could be from chemicals used to clean the instruments before departure from Earth. The most commonly used detection technique employed by these missions is to scoop or drill a sample, bring it back into the lander or rover and then heat it in an inert atmosphere and see what gases come off. This technique is called pyrolysis and is widely used on Earth to analyse the minerals and organic compounds within samples. Heat decomposes the materials and the gaseous fragments can be analysed and used to work out the type of compounds that were in the sample. A key requirement for successful pyrolysis is an inert atmosphere, a potential problem for Mars missions is whether the minerals introduced alongside potential organic molecules remain stable at the high temperatures the sample is heated to. Most minerals will remain inert, however some will release carbon dioxide and water and the most problematic will release oxygen. Oxygen can combust organic compounds so that all that is detected is carbon dioxide and water.
True to Mars’ track record of making exploration of its surface as complicated as possible it just so happens that it is coated in a mineral that releases oxygen on heating. The mineral is a salt of chlorine and oxygen called perchlorate. It was first discovered by the Phoenix Lander in the polar regions of Mars in 2008 and has also been detected by Curiosity Rover in Gale Crater. It’s extremely rare on Earth, where it is only seen naturally in extremely arid environments such as the Atacama Desert or the Dry Valleys of Antarctica. Perchlorates also release chlorine on heating and it is possible that they are responsible not just for unsuccessful organic detection experiments but the fact we see simple chlorinated organic compounds when we heat Martian samples.
However, this may not be a complete disaster. Carbon dioxide is easy to analyse and the temperature at which it is detected could potentially indicate the nature of the material the carbon in the carbon dioxide is sourced from. New research has highlighted the fact that if perchlorate is heated with low weight organic matter (typical of contamination) you get a lower temperature carbon dioxide peak than if you heat perchlorate with a mix of organic matter weights (typical of indigenous organic matter). Chlorinated carbon compounds, like those seen by Viking and Curiosity, were also detected during these heating experiments.
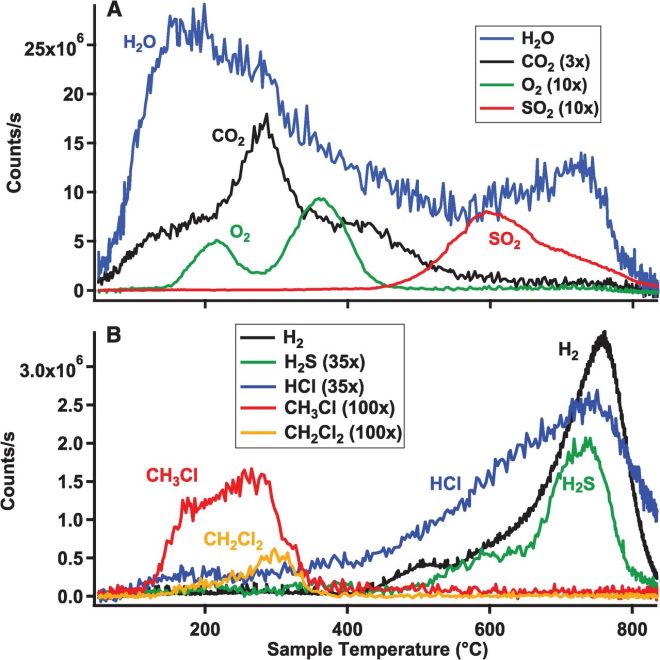
The release of gases at different temperatures from the Sheepbed mudstone sample during analysis by Curiosity Rover. Figure from Ming et al. 2014.
When Curiosity Rover heated a sample from within Gale Crater called the Sheepbed mudstone oxygen and chlorinated carbon compounds were seen to be released at the same temperature. This indicates that perchlorate sourced oxygen and chlorine were interacting with organic matter but the low temperature of releases suggests that in this case it was just contamination. Though searching for organic compounds on Mars has been found to be extremely difficult some information can still be gained from heating experiments. As we approach an era where sample return missions (that is sending a mission to Mars that then sends back samples to Earth) or even manned missions are likely, having an understanding of where on Mars may be organic rich or which rocks units may be most promising will be extremely valuable.
I was a co-author on the new pyrolysis of perchlorate and different molecular weights of organic matter research and you can get the open access paper by clicking here.
Please post any comments and questions below or to my twitter account.

